Blocking an enzyme linked to the immune response shows promise for preventing or treating severe symptoms of COVID-19 by reducing inflammation, tissue damage and blood clots in the lungs, new research in mice shows.
Scientists who have long studied the functions of this molecule in bacterial infections have traced the development of extensive lung lesions in infected mice to increased levels of the enzyme triggered by the invading SARS-CoV-2 virus.
Versions of this enzyme exist and perform similar functions in mice and humans - they are called caspase 11 and caspase 4, respectively. After discovering that the molecule is an attractive therapeutic target, researchers are exploring compounds that could safely and effectively block its activation.
Amer teamed up with Ohio State flu and COVID virologist Jacob Yount to examine the role of caspase 11 in coronavirus infection. Their labs have conducted a number of experiments comparing the results of COVID infection in normal mice and mice genetically modified so that they do not produce the enzyme.
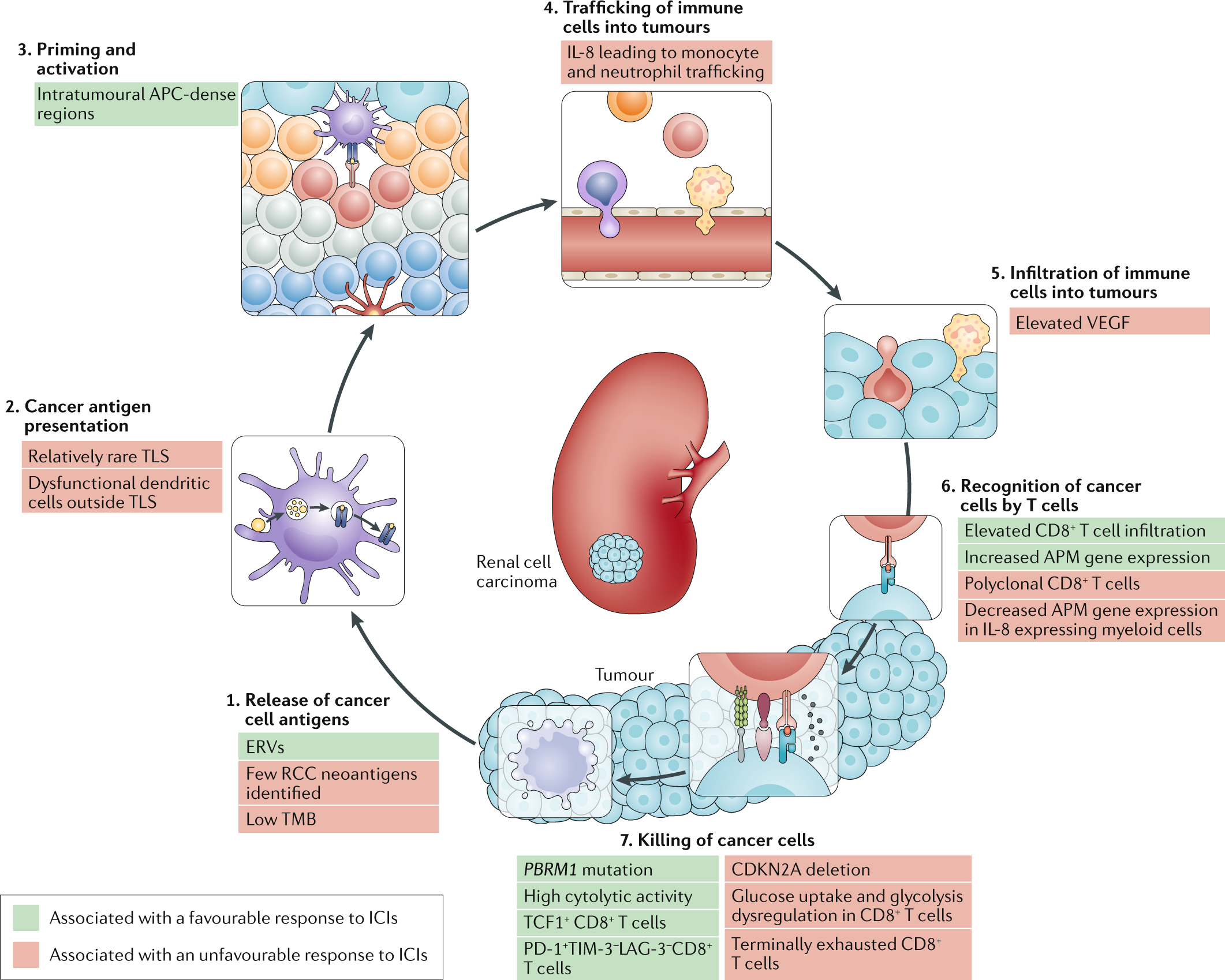
“From the first experiment, we saw that the caspase 11 knockout mice had less severe infections and started to recover after just a few days,” said Yount, associate professor of microbial infection and immunity. and co-lead author of the study.
Previous research has shown that caspase 11 in mice has many of the same immune response functions as caspase 4 in humans. In both species, the enzyme is produced at the start of an infection.
This study supports the idea that what was observed in mice is relevant to humans: researchers analyzed COVID-19 patient data available nationwide and found that caspase 4 was highly expressed in mice. people hospitalized in the ICU - linking its presence to a serious illness. Lung tissue samples from COVID patients also showed high activation of the molecule.
The sickest COVID-19 patients develop acute respiratory distress syndrome resulting from the combination of high levels of pro-inflammatory proteins called cytokines, fluid buildup in the air sacs that seeps into lung tissue and blood clots, or thrombosis, caused by damage to the cells lining the vessel walls.
In a series of experiments, the research team found that inhibiting caspase 11 reduced the intensity of multiple effects. They used a version of the SARS-CoV-2 virus that other scientists engineered specifically to cause disease in mice. (The human coronavirus does not make mice sick.)
Among the most striking results: lower recruitment and inflammatory priming of first responder cells called neutrophils, white blood cells whose job is to heal wounds and clear infection - they are important, but tend to perpetuate inflammation that damages tissue and contributes to blood clots. A technique used to image the smallest capillaries in the lungs of mice also showed that while the blood vessels in the lungs of normal mice infected with the virus were speckled with clots, the capillaries of mice lacking caspase 11 remained free of thrombosis.
"What happens in the lungs with COVID may be worse than with other infections. It's amazing that caspase 11 controls many of these unique aspects of COVID-19 pathology," Yount said.
Amer said this research has opened up new ways of thinking about the enzyme's possible role in a host of diseases. Its role in worsening lung damage in COVID-19 was an unexpected finding – activation of caspase 11 and caspase 4 in bacterial infections has a protective function, setting up immune cells to kill agents bacterial pathogens.
Caspase 11 is known to need help from a specific protein called gasdermin-D to ward off bacterial infections, but this work showed that the enzyme intensified lung damage in COVID-19 infection without using by gasdermin-D. The link to blood clotting also suggests that caspase 11's effects in the presence of an infection likely don't stop at the lungs and can affect diseases of the heart, brain, and elsewhere in the body.
